
The FDA has approved nivolumab (Opdivo) for patients with unresectable or metastatic melanoma following treatment with ipilimumab or a BRAF inhibitor, based on data from the phase III CheckMate-037 trial.

The FDA has approved nivolumab (Opdivo) for patients with unresectable or metastatic melanoma following treatment with ipilimumab or a BRAF inhibitor, based on data from the phase III CheckMate-037 trial.

Edith A. Perez, MD, the deputy director at large for the Mayo Clinic Cancer Center, comments on immune checkpoint inhibition for the treatment of breast cancer.

Sara Hurvitz, MD, medical oncologist, UCLA Medical Center, discusses the results of a study looking at pembrolizumab (Keytruda) in patients with triple-negative breast cancer (TNBC).

Jae Park, MD, assistant attending physician, Memorial Sloan Kettering Cancer Center, discusses CAR T-cell therapy for the treatment of acute lymphoblastic leukemia.

The anti-CD19 chimeric antigen receptor (CAR)-modified T-cell therapy CTL019 demonstrated a 92% complete response (CR) rate in pediatric patients with relapsed/refractory acute lymphoblastic leukemia (ALL).

Most patients with classical Hodgkin lymphoma (cHL), having previously failed three or more therapies, responded to the immunotherapy nivolumab in a small phase I trial.

Treatment with the PD-1 inhibitor pembrolizumab (Keytruda) elicited responses in 66% of patients with classical Hodgkin lymphoma (cHL).

Philippe Armand, MD, PhD, senior physician, Dana-Farber Cancer Institute, discusses the utility of PD-1 inhibitors for hematologic malignancies.

Pfizer and Merck KGaA will collaborate on the development of the PD-L1 inhibitor MSB0010718C as a potential treatment for multiple types of cancer, according to the companies.

The programmed death receptor-1 (PD-1) ligand, PD-L1, has become a viable target for immunotherapy in cancer, with multiple antibodies now in development.

Clinicians will soon face challenging decisions on which immunotherapies to prescribe their patients, and in what sequence or combination.

Multiple trials are ongoing in NSCLC with immunotherapy agents. As with most cancers, however, there is also a need to identify which types of patients with NSCLC might benefit the most from these new therapies.

Vassiliki Papadimitrakopoulou, MD, discusses adverse events associated with immunotherapies for the treatment of lung cancer.

In malignancies with limited response rates to existing radiation and/or chemotherapy, immunotherapy is being investigated as a potential adjunct, with promising results, particularly in the treatment of malignant mesothelioma.

Barbara Burtness, MD, discusses potential immunotherapy agents that may assist in the treatment of head and neck cancers.
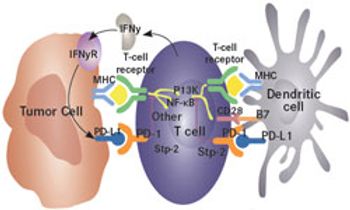
It is estimated that 1 in 63 individuals in the United States will develop renal cell carcinoma (RCC), making it among the most common cancers in the country.

William K. Oh, MD, discusses immunotherapies for the treatment of patients with prostate cancer.
The FDA has granted the chimeric antigen receptor (CAR) T cell therapy JCAR015 a breakthrough therapy designation as a treatment for patients with relapsed or refractory B-cell acute lymphoblastic leukemia (ALL).
Checkpoint inhibition has demonstrated efficacy for the treatment of melanoma in several clinical trials. However, biomarkers to predict safety and efficacy of immunotherapies in individual melanoma patients are lacking.

Ongoing clinical trials are evaluating a new immunotherapeutic approach for the treatment of malignant melanoma–the combination of an oncolytic virus with checkpoint inhibition.

Scientists are beginning to find that combination therapy improves outcomes for patients, particularly with ipilimumab-nivolumab combination therapy.
As its CAR T cell and high-affinity TCR products continue to advance in clinical trials, Juno Therapeutics, Inc, filed a registration statement for an initial public offering (IPO) of its common stock on November 17.

Pembrolizumab (Keytruda) significantly improved progression-free survival (PFS) by over 43% compared with chemotherapy as a treatment for patients with metastatic melanoma who were refractory to ipilimumab (Yervoy).

Frontline treatment with nivolumab (Opdivo) significantly extended overall survival (OS) and progression-free survival (PFS) when compared with dacarbazine in patients with untreated BRAF wild-type advanced melanoma.

Several novel approaches to the delivery of therapeutics for advanced melanoma may improve treatment efficacy and patient survival.