Melanoma
Latest News
Latest Videos
CME Content
More News

Patients with BRAF-mutant late-stage melanoma derive a lasting overall survival (OS) benefit from treatment with the combination of dabrafenib and trametinib compared with dabrafenib and placebo, according to the final results from the COMBI-d phase III double-blinded trial.

Treatment with the PD-1 inhibitor nivolumab demonstrated similar efficacy regardless of prior treatment with a BRAF inhibitor or ipilimumab in patients with BRAF mutant or wild type metastatic melanoma.
The PD-1 inhibitor nivolumab (Opdivo) has been has been assigned priority review designation from the US Food and Drug Administration (FDA) as a treatment for previously untreated patients with unresectable or metastatic melanoma.

Mark R. Middleton, MD, PhD, discusses the results of a phase I/IIa trial of IMCgp100, a bispecific immunotherapy, for the treatment of patients with melanoma.

Results from the phase II, double-blind CheckMate-069 clinical trial showed unprecedented response rates with ipilimumab combined with nivolumab in previously treated patients with metastatic melanoma.

Jeffrey S. Weber, MD, PhD, a senior member at Moffitt Cancer Center, discusses toxicities associated with immunotherapies for the treatment of patients with melanoma.

Ahmad Tarhini, MD, PhD, associate professor, University of Pittsburgh School of Medicine, compares two doses of ipilimumab (Yervoy).
Pembrolizumab (Keytruda) yielded significantly better outcomes compared with ipilimumab (Yervoy) in a randomized phase III trial of patients with advanced melanoma.

IMCgp100, an anti-CD3 antibody fragment fused to a gp100-specific T cell receptor, yielded long-lasting responses in patients with advanced melanoma.
A delay between the diagnosis of melanoma and needed surgery may evoke anxiety and psychological stress; although the impact on morbidity and mortality remains controversial.
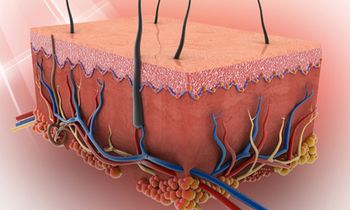
Abnormal driver mutations contribute to tumor progression and have been prime targets for many therapeutic studies in oncology; however, researchers are focusing more and more on the less-studied passenger mutations and their role in tumor progression.

Despite the availability of several new agents in the past 5 years for the treatment of melanoma, patients with advanced melanoma still suffer poor prognoses.

Kevin B. Kim, MD, medical oncology, California Pacific Medical Center, discusses the outlook for T-VEC for the treatment of patients with melanoma.
Turning their experimental focus on the tumor microenvironment, the authors of a paper published in Clinical Cancer Research have shed light on the role of melanoma-associated tumor macrophages in resistance to BRAF V600E inhibitors.
According to the Skin Cancer Foundation, more than 3.5 million cases of nonmelanoma skin cancers (NMSCs) are treated in the United States annually, making it the most commonly encountered malignancy.

Mario E. Lacouture, MD, dermatologist, Memorial Sloan Kettering Cancer Center, discusses the impact on quality of life of dermatologic adverse events.

A trial comparing frontline pembrolizumab with ipilimumab for the treatment of advanced melanoma has met its progression-free survival (PFS) and overall survival (OS) endpoints and will be stopped early.

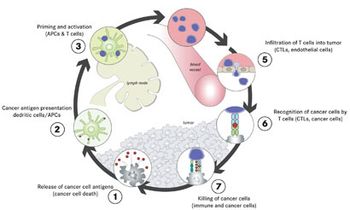
Immunotherapy is a rapidly expanding approach to the treatment of melanoma, employing a number of strategies evident in the pipeline for immunotherapeutics.
Melanoma is the most deadly skin cancer. According to data collected between 2004 and 2010, only 16% of Americans with metastatic melanoma, including people of all ages and races, and both genders, survive for 5 years or more after diagnosis.

Jason J. Luke, MD, assistant professor of medicine, The University of Chicago, discusses PD-1 inhibitors for the treatment of patients with melanoma.

Omid Hamid, MD, discusses sequencing and combining targeted therapies in the treatment of melanoma.
In an analysis of adverse events following treatment of patients with advanced melanoma with ipilimumab and nivolumab, combination therapy was associated with a 22% incidence of either thyroiditis or hypothyroidism and a 9% incidence of hypophysitis.

The FDA has accepted a supplemental Biologics License Application (sBLA) for ipilimumab (Yervoy) as an adjuvant treatment of patients with stage III melanoma at high risk of recurrence following complete resection.

The MEK inhibitor cobimetinib has received an FDA priority review for use in combination with the BRAF inhibitor vemurafenib (Zelboraf) to treat patients with BRAF V600–positive advanced melanoma.

An international team of scientists has shed additional light on the important role of Yes-associated protein (YAP) in tumor development and in treatment response.