
BREAST CANCER
Latest News
Latest Videos

CME Content
More News
The next-generation PI3 kinase (PI3K) inhibitor GDC-0032 has shown signs of efficacy in patients with advanced cancers that were mutated for the PI3K alpha gene.

Emiel J. Rutgers, MD, from the Netherlands Cancer Institute, describes the personalization of treatment for patients with early-stage breast cancer.
If approved, this regimen would be the first neoadjuvant regimen ever approved for the treatment of cancer.

Jane E. Churpek, MD, describes the BROCA genetic assay, uses targeted genomic capture and next-generation sequencing to analyze a panel of 42 genes.
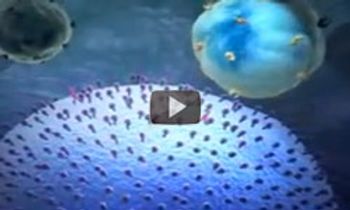
The mechanism of action of trastuzumab (Herceptin), a monoclonal antibody that is approved for the treatment of early-stage breast cancer that is HER2-positive.

Richard G. Gray, MA, MSc, discusses results from the randomized, phase III aTTom study that examined 10 years of tamoxifen versus the more standard 5 years.

Richard Finn, MD, comments on the future of palbociclib (PD 0332991), a novel oral selective inhibitor of cyclin-dependent kinase 4/6 (CDK 4/6), for the treatment of breast cancer.

Data from the French National Cancer Institute showed an increase in testing for BRCA1/2 for breast and ovarian cancer, though not for the MMR mutation for Lynch syndrome.

Andrew D. Seidman, MD, comments on the findings of the phase III S0221 trial, which compared two schedules of paclitaxel as adjuvant therapy for breast cancer.

Mark D. Pegram, MD, describes research into the antibody-drug conjugate T-DM1 (trastuzumab emtansine), following its FDA approval in February as a treatment for HER2-positive metastatic breast cancer.

Ado-trastuzumab emtansine received FDA approval in February, and a number of ongoing trials now seek to determine whether the antibody-drug conjugate can be combined with other agents.

George W. Sledge, Jr, MD, Chief, Division of Oncology, Professor of Medicine, Stanford University School of Medicine, discusses the mechanisms of resistance in breast cancer.

The American Association for Cancer Research is an annual showcase of the latest scientific cancer research, with updates on phase I, often first-in-human clinical trials.

Lisa A. Carey, MD, Professor of Medicine, University of North Carolina at Chapel Hill, discusses assays and intrinsic subtypes of breast cancer.

Fabrice Andre, MD, PhD, says that there are no targets in triple-negative breast cancer in accordance with the current vision of the disease.

Debu Tripathy, MD, Co-Leader, Women's Cancer Program, Norris Comprehensive Cancer Center, University of Southern California, describes intratumoral heterogeneity in patients with breast cancer.

Martine Piccart, MD, PhD, director of medicine at the Jules Bordet Institute in Brussels, Belgium, discusses the changing view of anti-HER2 therapies for patients with breast cancer.

Debu Tripathy, MD, Co-Leader, Women's Cancer Program, Norris Comprehensive Cancer Center, University of Southern California, discusses two important trials in breast cancer looking at extended hormonal therapy.

José Baselga, MD, PhD, Physician-in-Chief, Memorial Sloan-Kettering Cancer Center, discusses targeting the phosphoinositide 3-kinase (PI3K) in breast cancer.

Lisa A. Carey, MD, Professor of Medicine, University of North Carolina at Chapel Hill, discusses the challenges of triple-negative breast cancer.

Fabrice Andre, MD, PhD, Research Director, Head of INSERM Unit U981, Institut National des Sciences et de la Recherche Médicale, Villejuif, France, discusses the need for new testing methods.

Martine Piccart, MD, PhD, director of medicine at the Jules Bordet Institute in Brussels, Belgium, discusses genomic testing in breast cancer.

The 30th Annual Miami Breast Cancer Conference (MBCC) at the Fontainebleau Miami Beach Hotel, in Miami Florida, from March 7-10, 2013.

Approximately 20% of human breast cancers are characterized by overexpression of the HER2 receptor tyrosine kinase molecule. Multiple anitbodies have been designed to specifically target HER2, and each is able to target the molecule at a different epitope.

George W. Sledge, Jr, MD, Chief, Division of Oncology, Professor of Medicine, Stanford University School of Medicine, discusses drug toxicity among targeted agents.










































