
Bladder Cancer
Latest News
Latest Videos

CME Content
More News

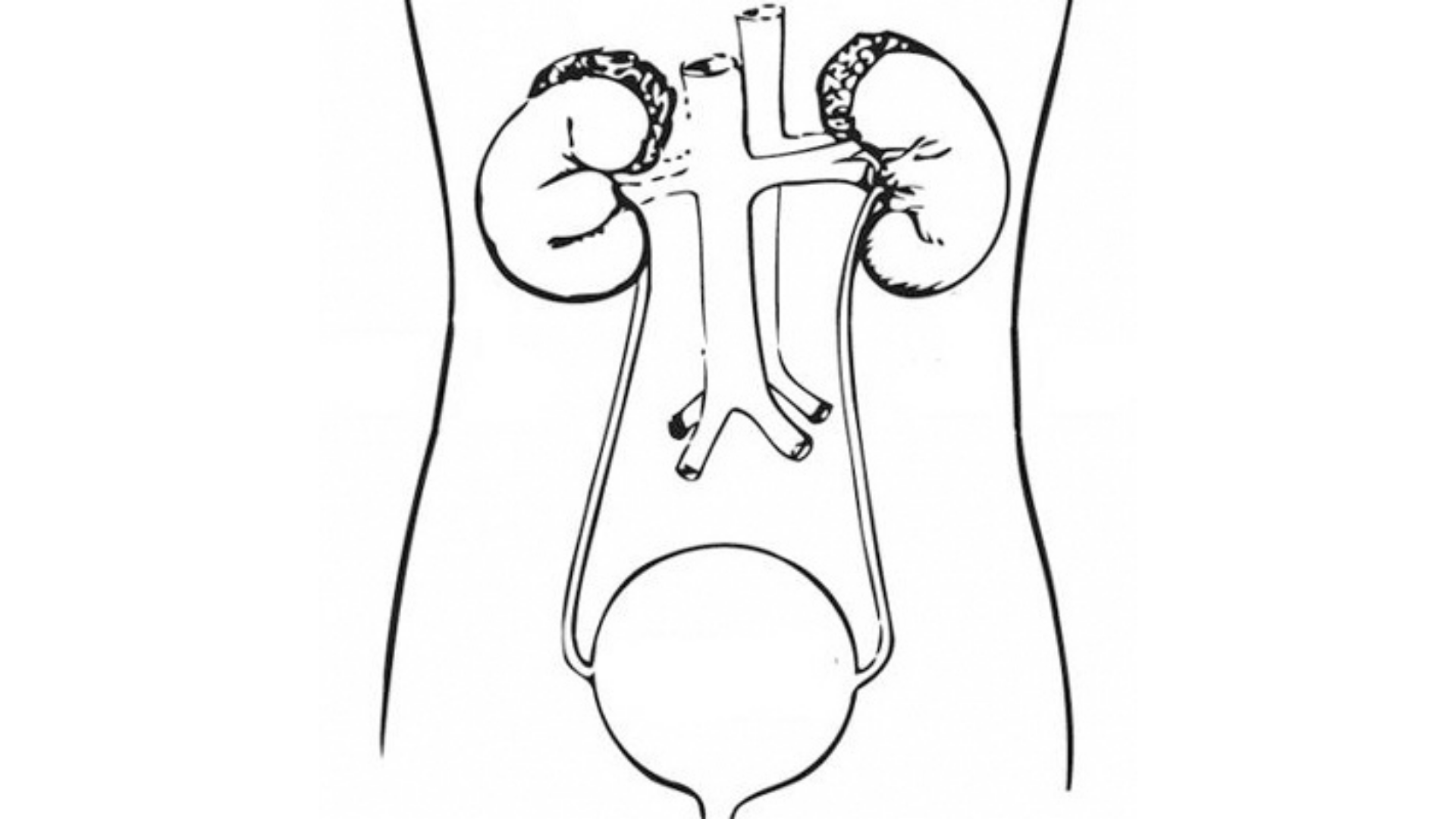
TAR-200 is being studied for carcinoma in situ with or without papillary disease since it addresses diffuse CIS that cannot be surgically removed, though future trials will expand to papillary disease as adjuvant therapy like BCG, with the goal of extending the disease-free period before potentially reintroducing therapy as needed.

The TAR-200 treatment has demonstrated a favorable safety profile in clinical trial, with primarily low-grade irritated voiding symptoms that are manageable and expected with intravesical therapy, without concerning systemic side effects, making it a promising new option to delay or reduce radical cystectomy in patients with BCG-unresponsive NMIBC.

The TAR-200 treatment for BCG-unresponsive NMIBC works through a catheter-placed intravesical device that elutes gemcitabine over 3 weeks, providing a high complete response rate of 77% in early testing, likely due to the sustained release mechanism.

The FDA has granted 2 designations to cretostimogene grenadenorepvec, an oncolytic immunotherapy, for the treatment of a specific type of high-risk bladder cancer:

TAR-200, a novel targeted therapy, has been granted breakthrough therapy designation by the FDA in high-risk non-muscle-invasive bladder cancer that is unresponsive to Bacillus Calmette-Guérin.

Summary: The FDA has accepted for priority review nivolumab and cisplatin-based chemotherapy as a first line of treatment in patients with inoperable or metastatic urothelial carcinoma.
Findings presented during the 2023 ESMO Annual Meeting have demonstrated a new combination of targeted therapies offering clinicians and patients renewed hope in this difficult to treat cancer type.

Positive data from the phase 3 EV-302 trial of enfortumab vedotin with pembrolizumab in locally advanced or metastatic urothelial cancer have shifted the landscape.

BCG-unresponsive NMIBC treatment aims to eradicate disease or prolong disease-free intervals, with radical cystectomy as gold standard; newer options like pembrolizumab and gemcitabine/docetaxel provide improved alternatives, though logistic challenges remain.

BCG-unresponsive non-muscle invasive bladder cancer risks invasive progression; providers should suspect disease based on any abnormalities in cystoscopy and cytology, not just voiding symptoms.

An analysis, based on data from the JAVELIN 100 Bladder trial, found the NCCN/FACT Bladder Symptom Index-18 for assessing quality-of-life in patients with advanced urothelial carcinoma to be reliable and valid.

With an investigational new drug granted to ZH9 by the FDA, a clinical development program for the treatment of patients with non-muscle invasive bladder cancer can be initiated.

The combination of sacituzumab govitecan-hziy and enfortumab vedotin-ejfv showed early responses in patients with treatment-resistant metastatic urothelial cancer.

In an interview with Targeted Oncology, Brian Ramnaraign, MD, further discussed the safety of immune checkpoint inhibitors in patients with genitourinary cancers.

After issuing a complete response letter in May 2023, the FDA has now accepted the resubmission of the biologics license application of a drug regimen for the treatment of non-muscle-invasive bladder cancer.

A phase 2 study of neoadjuvant gemcitabine, cisplatin, plus nivolumab met its co-primary end point with a positive predictive value of clinical complete response of 0.97 among patients with muscle-invasive bladder cancer.

Over three-fourths of patients with BCG-unresponsive, high-risk non–muscle-invasive bladder cancer treated with TAR-200 achieved complete responses in the phase 2b SUNRISE-1 trial.

Enfortumab vedotin-ejfv plus pembrolizumab improves survival in patients with previously untreated locally advanced or metastatic urothelial carcinoma.

The addition of nivolumab to frontline standard of care gemcitabine-cisplatin, followed by nivolumab maintenance therapy, led to statistically significant and clinically meaningful improvements in survival compared with gemcitabine-cisplatin alone in patients with unresectable or metastatic urothelial carcinoma.

A post hoc analysis of overall survival and progression-free survival support the use of avelumab with best supportive care as a standard of care for patients with urothelial carcinoma.

Findings from the CheckMate-901 trial demonstrated improvements for the treatment of patients with urothelial carcinoma who received a combination of nivolumab and chemotherapy followed by nivolumab monotherapy.

Meeting a primary end point, KEYNOTE-123 study shows promise for pembrolizumab as an adjuvant muscle-invasive bladder cancer treatment.

The ADVANCED-2 trial plans to build onto the early antitumor and safety data seen with TARA-002 that were presented this year for patients with non-muscle invasive bladder cancer.
Seagen Inc., and Astellas Pharma Inc., report that both primary end points of the phase 3 EV-302 clinical trial have been met.

In an interview with Targeted Oncology, Arlene O. Siefker-Radtke, MD, discusses the findings from 3 trials of erdafitinib and explains the next steps for evaluating the agent.
































