
Lanreotide (Somatuline Depot) improved progression-free survival and resulted in more disease control compared with an observation strategy among patients with pancreatic neuroendocrine tumors (pNETs).

Lanreotide (Somatuline Depot) improved progression-free survival and resulted in more disease control compared with an observation strategy among patients with pancreatic neuroendocrine tumors (pNETs).

Amgen and Kite Pharma have announced that they will collaborate on the development of novel CAR T-cell immunotherapies, with Amgen providing cancer targets and Kite offering its engineered autologous cell therapy platform.

A 12-gene test for breast cancer recurrence after ductal carcinoma in situ (DCIS) distinguished high- and intermediate- risk patients from those with a low risk. These results were presented at the 2014 San Antonio Breast Cancer Symposium.

The anti-CD19 chimeric antigen receptor (CAR)-modified T-cell therapy CTL019 demonstrated a 92% complete response (CR) rate in pediatric patients with relapsed/refractory acute lymphoblastic leukemia (ALL).
As its CAR T cell and high-affinity TCR products continue to advance in clinical trials, Juno Therapeutics, Inc, filed a registration statement for an initial public offering (IPO) of its common stock on November 17.

Recent news stories profiling a cancer patient whose last hope rests on treatment by injections of the virus that causes AIDS may have created some misconceptions regarding a new cancer immunotherapy.

Based on data from the phase III RAINBOW trial, the FDA has approved ramucirumab (Cyramza) in combination with paclitaxel as a treatment for patients with previously treated advanced gastric or gastroesophageal junction (GEJ) adenocarcinoma.

William D. Tap, MD, discusses the potential to combine KIT and CTLA-4 blockade in patients with refractory GIST and other advanced sarcomas.

A head-to-head comparison of cetuximab and bevacizumab showed equivalence for chemotherapy plus either agent in terms of OS, PFS, and response rates for patients with certain previously untreated mCRC.

Tanios Bekaii-Saab, MD, Section Chief, Gastrointestinal Oncology, associate professor, Internal Medicine, Pharmacology, The Ohio State University, discusses an analysis of two targeted drugs for the treatment of patients with colorectal cancer (CRC).

Last week, the US Food and Drug Administration (FDA) announced two steps it will take to ensure the safety and utility of certain diagnostic tests.

The investigational CD19-targeted chimeric antigen receptor (CAR) therapy CTL019 has received a breakthrough therapy designation from the FDA as a potential treatment for pediatric and adult patients with relapsed/refractory acute lymphoblastic leukemia (ALL).

Raoul S. Concepcion, MD, FACS, Urology Associates P.C., discusses the potential for the 4Kscore test as a predictor of high-grade prostate cancer.

The FDA has approved the radioactive diagnostic imaging agent Lymphoseek injection to guide sentinel lymph node biopsy in patients with cancer of the head and neck.

A cell-cycle gene array test demonstrated independent value for predicting metastatic progression after surgery for organ-confined renal cell carcinoma (RCC) of clear cell histology.

Ghassan K. Abou-Alfa, MD, Gastrointestinal Oncology Service, Memorial Sloan Kettering Cancer Center, discusses a retrospective study to determine the prognostic value of C-reactive protein (CRP) levels in patients with hepatocellular carcinoma (HCC) who are undergoing treatment with sorafenib.
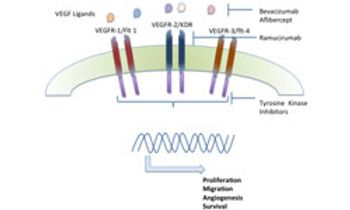
Advanced gastric cancer and gastroesophageal junction cancers are highly aggressive cancers with poor prognosis and limited treatment options. The VEGF pathway has been one of the key signaling pathways under investigation for these cancers.

Marcel R.M. van den Brink, MD, PhD, from Memorial Sloan Kettering Cancer Center, describes a novel CLIA-certified next-generation sequencing-based assay for hematologic malignancies.
The FDA has approved ramucirumab as a treatment for patients with unresectable gastric cancer or GEJ adenocarcinoma following fluoropyrimidine- or platinum-containing therapy, based on a significant extension in overall survival (OS).

Ghassan K. Abou-Alfa, MD, Gastrointestinal Oncology Service, Memorial Sloan-Kettering Cancer Center, discusses the phase II study of a novel transforming growth factor-beta receptor I (TGF-β1) kinase inhibitor, LY2157299 monohydrate, in patients with advanced hepatocellular carcinoma (HCC), which was presented at the 2014 Gastrointestinal Cancers Symposium

Screening for prostate-specific antigen (PSA) significantly cuts the death rate from prostate cancer, but at the same time, America’s medical community should work harder to avoid the screen’s potential pitfalls.

A wide-ranging analysis of more than 5500 breast cancer tumors that combined genomic and protein expression testing has identified promising targets to explore for treating patients with poor prognoses, with particularly notable findings involving androgen receptor (AR) expression.

Sukumar Nagendran, MD, vice president, medical affairs, Quest Diagnostics, describes BRCAvantage, a test to detect BRCA1 and BRCA2 genes.

Vicki Keedy, MD, Assistant Professor of Medicine, Clinical Director, Sarcoma Program, Vanderbilt-Ingram Cancer Center, discusses novel agents that are currently under investigation for the treatment of gastrointestinal stromal tumors

Julie R. Brahmer, MD, from Johns Hopkins University School of Medicine, Sidney Kimmel Comprehensive Cancer Center, discusses the outlook for immunotherapies in cancer care.