
With the emergence of antibody-drug conjugates, the genitourinary cancers space is rapidly evolving. Dr Ferris addresses practice-changing presentations from the 2026 ASCO GU Cancers Symposium.
Robert L. Ferris, MD, PhD, is a head and neck surgical oncologist with an NIH R01 funded basic/translational immunology laboratory. He is the director of the University of Pittsburgh Medical Center Hillman Cancer Center, a Hillman professor of oncology, associate vice chancellor for cancer research, codirector of the Tumor Microenvironment Center, and a professor in the Department of Otolaryngology, of Immunology, and of Radiation Oncology.

With the emergence of antibody-drug conjugates, the genitourinary cancers space is rapidly evolving. Dr Ferris addresses practice-changing presentations from the 2026 ASCO GU Cancers Symposium.

A wave of innovations across antibody-drug conjugates, synergistic combinations, and expanding biomarkers is transforming the genitourinary cancer landscape from metastatic rescue toward curative, earlier-line paradigms.

AI revolutionizes cancer care by enhancing detection, diagnosis, and personalized treatment, promising improved outcomes and efficiency in oncology.


Robert L. Ferris, MD, PhD, editor-in-chief of Targeted Therapies in Oncology, discusses his monthly column in this episode of Chief Insights in Oncology.

Robert L. Ferris, MD, PhD, pens this month's editor-in-chief column for Targeted Therapies in Oncology.
Perioperative pembrolizumab enhances survival rates in advanced head and neck cancer, paving the way for earlier immunotherapy integration and future treatment strategies.

The field of cancer research and treatment is, almost by definition, continually evolving.

The GI Cancers Symposium revealed new therapies for specific cancer subsets and progress in chimeric antigen receptor T-cell therapies for solid tumors.

Given different scientific and clinical meeting schedules, considering advances in genitourinary oncology in novel therapies is relevant.

The advent of cellular therapies, such as chimeric antigen receptor T-cell therapy, has revolutionized the field of hematologic malignancies.

The 2024 Nobel Prize in Chemistry highlights AI-driven breakthroughs in computational protein design and structure prediction. Tools like AlphaFold promise faster development of targeted cancer drugs and transformative advances in oncology and medicine.

Recent advancements in protein engineering, especially antibody-drug conjugates, show promise in lung cancer treatment, with ivonescimab outperforming pembrolizumab in PD-L1-positive advanced non-small cell lung cancer.
Robert L. Ferris, MD, PhD, highlights groundbreaking advancements presented at the recent World Congress on Lung Cancer held in San Diego, California.

Cancer treatment is advancing through the use of tumor-targeted antibodies combined with toxic payloads like chemotherapy agents and radioisotopes.

The debate on non-specific vs targeted therapies in oncology highlights the need for a balanced approach optimizing existing agents while developing biomarkers and precision medicine.

Due to federal budget constraints, the National Cancer Institute faces significant risk, impacting cancer research funding amid crucial breakthroughs.

Until recently, the CAR gene modification has been focused on T cells, thought for decades to be the main effector population for controlling cancer or for harnessing cancer immunotherapy.

It is a promising time for cellular therapy, and combinations with gene modification or other systemic or local therapies bode well for solid tumor development of this TIL therapy platform.

The growing number of clinical trials testing neoadjuvant immunotherapy with or without chemotherapy has opened up new possibilities for treating locally advanced solid tumors.

The recent announcement of the 2023 Nobel Prize in Physiology or Medicine has turned attention again to RNA therapeutic vaccines and a better understanding of RNA biology.

The classes of cellular immunotherapies and those blocking negative regulatory signals, known as immune checkpoint blockade, have revolutionized cancer treatment.

Robert L. Ferris, MD, PhD, discussed the growing use of bispecific antibodies and how he expects to see more of these advances in the coming years.

Robert L. Ferris, MD, PhD, discusses what should be done when a trial results in a negative finding.

Robert L. Ferris, MD, PhD, reflects on the status of telehealth in oncology and patient care following the COVID-19 pandemic.

the tremendous scientific developments in ctDNA, including genetic and epigenetic alterations, which are detectable in the blood—the so-called liquid biopsy—or multicancer early detection tests, represent a revolution that seems to be rapidly emerging on the horizon.
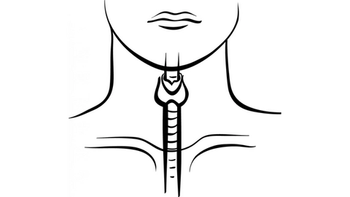
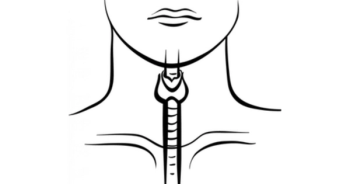
Advances in thyroid cancer to date hold promise for other orphan cancers with analogous alterations, on which we might bring to bear agents that will have similar effects.

The oncology community has enjoyed success to a large degree through the existing NIH and NCI mechanisms. Will they ask how can they work with us in a concerted fashion to benefit new discoveries, novel translation in well-designed clinical trials, and eventual development of exciting, more effective cancer therapies for patients?

Emerging from a 2-year pandemic, the work-from-home revolution has led to the migration of staff to the pharmaceutical industry or contract research organizations.

Harnessing existing and otherwise safe antibody-targeting strategies and combining them with chemotherapy or other bioactive compounds or as a bispecific dual antibody moiety are an exciting strategy of protein engineering that is emerging and coming into its own in oncology.

August 22nd 2025

August 27th 2025

March 26th 2026